
Looking for a flexible side hustle that doesn’t require special skills or upfront investment? Participating in Fortrea clinical trials might be the opportunity you’ve been overlooking. As one of the world’s leading contract research organizations, Fortrea conducts thousands of clinical studies each year, and they’re always looking for healthy volunteers who can earn anywhere from a few hundred to several thousand dollars for their participation. Whether you’re between jobs, building your freelance career, or just want some extra cash on the side, clinical trials offer a unique way to make money while contributing to medical research that could help millions of people worldwide.
What Are Fortrea Clinical Trials and Why Do They Pay?
Fortrea clinical trials are research studies designed to test new medications, treatments, and medical devices before they can be approved for public use. Fortrea operates as a global contract research organization (CRO), working with pharmaceutical companies to conduct these essential studies across multiple therapeutic areas.
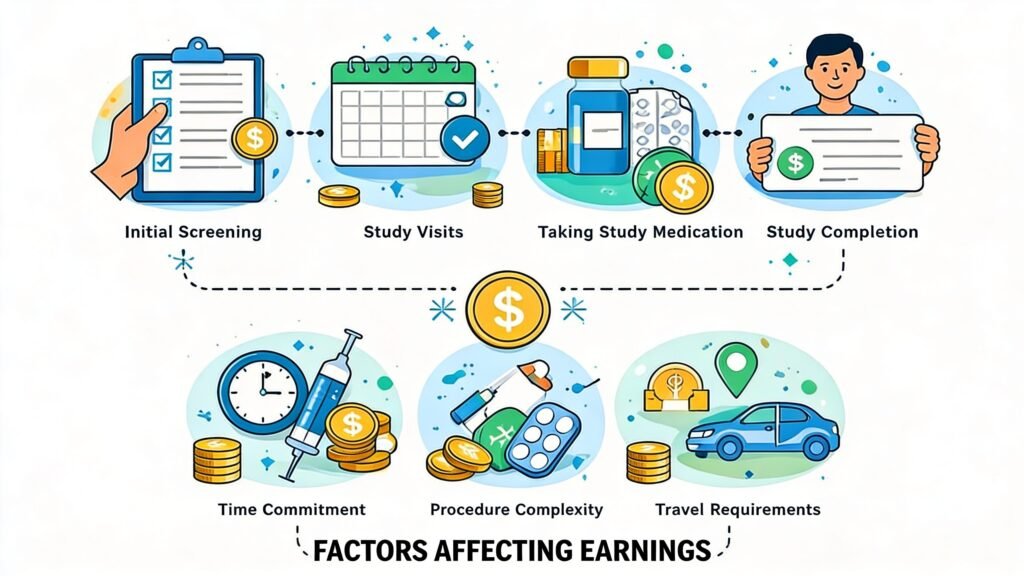
Here’s why they pay participants so well:
Time commitment: Studies can range from a few hours to several weeks, and your time has real value
Inconvenience factor: You might need to follow specific diet restrictions, stay overnight at a facility, or undergo multiple check-ups
Medical procedures: Some trials involve blood draws, imaging scans, or other procedures
Regulatory requirements: Companies need diverse participants to meet FDA guidelines and ensure medication safety
The compensation isn’t charity. It’s a legitimate payment for your valuable contribution to scientific advancement. Think of it as a side gig where you’re helping develop tomorrow’s breakthrough medications while padding your bank account today.
The Reality of Clinical Trial Compensation
Let’s talk numbers. Fortrea clinical trials typically pay anywhere from $50 for a simple screening visit to $10,000+ for longer inpatient studies. The amount you’ll earn depends on several factors:
Study Type | Duration | Typical Pay Range | Time Commitment |
|---|---|---|---|
Screening Only | 1-2 hours | $50-$150 | Single visit |
Outpatient Study | 2-4 weeks | $500-$2,000 | Multiple visits |
Inpatient Study | 3-7 days | $2,500-$5,000 | Overnight stays |
Long-term Study | 3-12 months | $5,000-$15,000+ | Regular check-ins |
Most participants receive payment after completing each phase of the study or at the end of the entire trial. Some studies pay via check, while others use prepaid debit cards or direct deposit.
How to Find and Apply for Fortrea Clinical Trials
Getting started with Fortrea clinical trials is easier than you might think. Fortrea operates facilities across the United States and maintains an extensive database of ongoing studies looking for participants.
Step 1: Visit the Fortrea Clinical Trials Website
Head over to Fortrea’s clinical research database where you can browse active studies. You’ll find a searchable interface that lets you filter by:
Your location (city and state)
Your age range
Gender requirements
Specific health conditions or healthy volunteer status
Study compensation amount
Step 2: Review Study Details
Each listing provides detailed information about what’s involved. For example, this healthy adult study shows clear eligibility criteria, duration, and what participants can expect.
Step 3: Submit Your Application
Most studies have an online pre-screening form. You’ll answer questions about:
Your age, weight, and height
Current medications and supplements
Medical history
Lifestyle habits (smoking, alcohol consumption, exercise)
Availability for the study dates
Step 4: Complete the Screening Process
If you’re a potential match, you’ll be contacted for an in-person screening. This typically includes:
Physical examination
Blood and urine tests
Medical history review
Review of study requirements and informed consent
Similar to finding the best side gigs, success with clinical trials comes from being proactive and applying to multiple opportunities.
Who Can Participate in Clinical Trials?
The beauty of Fortrea clinical trials is that they need all types of people. While some studies target specific health conditions, many are looking for healthy volunteers with no significant medical issues.
General Eligibility Requirements
Healthy Volunteer Studies:
Typically ages 18-65 (some studies go younger or older)
BMI within a specific range (usually 18-32)
No prescription medications for chronic conditions
No recent participation in other clinical trials (usually 30-90 day waiting period)
Non-smoker or light smoker
Able to commit to all study visits
Disease-Specific Studies:
Diagnosed with the specific condition being studied
May be on stable medications
Age ranges vary based on the condition
Willing to potentially try experimental treatments
Common Disqualifications
Be aware that certain factors might prevent you from participating:
Pregnancy or breastfeeding
Recent blood donation
Active infections or illnesses
Certain tattoos or piercings (recent ones may cause issues)
Drug or alcohol dependency
Specific allergies related to the study medication
Fortrea’s therapeutic areas include oncology, neuroscience, cardiovascular, and infectious diseases, among others. This diversity means there’s likely a study you could qualify for, regardless of your health status.
What to Expect During Your Clinical Trial Experience
Understanding the process helps you prepare and maximize your earning potential from Fortrea clinical trials. Here’s what a typical study experience looks like:
Before the Study Begins
Once you’re accepted, you’ll receive detailed instructions. These might include:
Dietary restrictions (no caffeine, alcohol, or specific foods)
Medication restrictions (including over-the-counter meds and supplements)
Exercise limitations
Sleep requirements
You’ll sign informed consent documents that explain every aspect of the study, including potential risks and side effects. Take this seriously and ask questions about anything you don’t understand.
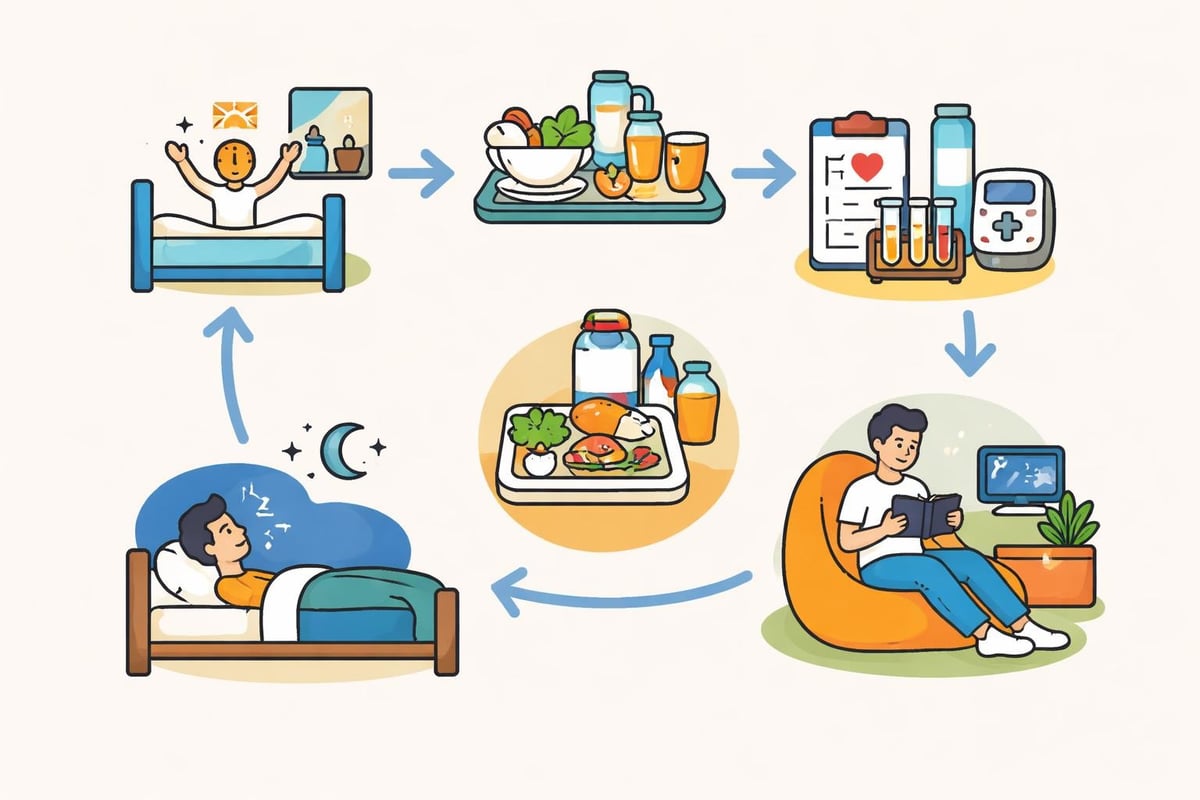
During Inpatient Studies
If you’re doing an inpatient study where you stay at the facility:
Check-in: Usually evening before the study officially starts
Baseline measurements: Vital signs, blood work, and health assessments
Study medication administration: You’ll receive the drug being tested or a placebo
Monitoring period: Regular blood draws, vital sign checks, and observations
Meals and activities: Provided meals, recreational time, and rest periods
Check-out: Final assessments before you leave
Think of it like getting paid to stay in a basic hotel where you eat provided meals, watch TV, read, and get your blood drawn a few times. Many participants bring laptops and catch up on work or starting a side hustle in 2026.
During Outpatient Studies
Outpatient studies require you to visit the facility periodically while living your normal life:
Weekly or monthly check-in appointments
Take study medication at home
Keep a diary of symptoms or side effects
Continue your regular routine between visits
More flexibility but requires diligent record-keeping
Maximizing Your Earnings from Clinical Trials
Treating clinical trial participation as a legitimate side hustle means approaching it strategically. Here’s how to maximize your income potential:
Join Multiple Study Databases
Don’t limit yourself to just Fortrea clinical trials. Register with multiple research organizations to increase your opportunities:
Local universities with research programs
Hospital-based research centers
Other CRO companies
Government-sponsored trials through clinicaltrials.gov
Track Your Study History
Keep detailed records of:
Which studies you’ve participated in
Dates of participation
Washout periods required
Compensation received
Contact information for coordinators
This helps you apply for new studies as soon as you’re eligible again. Most studies require a 30-90 day washout period between trials.
Build Relationships with Research Coordinators
Research coordinators remember reliable participants. If you:
Show up on time for all appointments
Follow study protocols exactly
Communicate clearly about any issues
Complete studies successfully
You’ll often get called first when new high-paying studies come available. It’s relationship building, similar to finding freelance clients.
Choose Higher-Paying Study Types
Study Characteristic | Impact on Compensation |
|---|---|
Longer duration | Higher total pay |
Overnight stays | Significant premium |
Multiple blood draws | Modest increase |
Dietary restrictions | Slight increase |
First-in-human trials | Premium compensation |
Placebo-controlled | Standard rates |
Safety Considerations and What You’re Really Signing Up For
Let’s be real about the risks involved in Fortrea clinical trials. While clinical trials are generally safe, you are taking experimental medications that haven’t been fully approved yet.
Understanding the Phases
Clinical trials happen in phases, each with different risk profiles:
Phase I Trials:
First time the drug is tested in humans
Usually highest compensation
Primarily focused on safety
Healthy volunteers typically used
Higher theoretical risk
Phase II & III Trials:
Drug has already been tested for safety
Now testing effectiveness
Often involve people with specific conditions
Lower risk profile
May offer less compensation
Phase IV Trials:
Drug is already approved
Post-market surveillance
Lowest risk
Often lower compensation
Real Talk About Side Effects
You might experience side effects ranging from minor to moderate:
Headaches
Nausea
Fatigue
Dizziness
Injection site reactions
Serious adverse events are rare because studies are closely monitored by medical professionals who can intervene immediately. Every study has stopping rules and safety protocols.
Medical Monitoring and Support
One underrated benefit of Fortrea clinical trials is the medical attention you receive:
Comprehensive health screening (often worth hundreds of dollars)
Regular monitoring by healthcare professionals
Immediate access to medical care if issues arise
Follow-up care if needed
Complete medical records of your participation
Many participants discover health issues during screening that they weren’t aware of, allowing them to seek treatment early.
How Fortrea Is Innovating the Clinical Trial Experience
Fortrea isn’t just running traditional clinical trials anymore. The company is investing heavily in technology to make participation easier and more efficient. Fortrea’s Intelligent Technology uses AI to streamline workflows and improve the participant experience.
What This Means for You as a Participant
Faster recruitment: Better matching means you hear back quicker
Reduced visit burden: Some assessments can be done remotely
Better communication: Automated reminders and updates
Improved diversity: Enhanced diversity initiatives mean more opportunities for underrepresented groups
These improvements make clinical trials more accessible as a side hustle option, requiring less time away from your other income streams.

Combining Clinical Trials with Other Side Hustles
The flexible nature of Fortrea clinical trials makes them perfect to combine with other income sources. Here’s how smart side hustlers stack their opportunities:
During Inpatient Studies
You’ve got hours of downtime while staying at the facility. Participants often:
Work on freelance projects on their laptops
Build passive income streams
Study for certifications
Write, code, or design
Manage online businesses
Just make sure your work doesn’t violate study protocols (like if you need to avoid screen time or stress).
Between Studies
The 30-90 day washout period between trials is perfect for:
Short-term freelance contracts
Building your client base
Trying different side hustles to diversify income
Creating a Rotating Income Strategy
Smart participants create a calendar that rotates between:
Clinical trial participation (big payment)
Washout period with active side hustles
Building skills or certifications
Next clinical trial opportunity
This approach generates consistent monthly income while avoiding burnout from any single source.
Tax Implications and Record Keeping
Here’s something most people don’t think about: clinical trial compensation is taxable income. Treat it like any other side hustle income for tax purposes.
What You Need to Know
Form 1099: You’ll receive a 1099 if you earn over $600 from a single study
Report all income: Even if you don’t get a 1099, you’re required to report the income
Self-employment tax: This isn’t W-2 income, so factor in self-employment taxes
Deductible expenses: Travel to and from the facility may be deductible
Keep These Records
Payment receipts or direct deposit confirmations
Study completion certificates
Travel logs and mileage
Any study-related expenses
1099 forms
Consider using the same tracking methods you’d use for pricing your freelance services to stay organized.
Common Questions and Misconceptions
“Is it safe to do multiple studies per year?”
Yes, as long as you respect washout periods and don’t overlap studies. Most healthy volunteers can safely participate in 3-4 studies annually.
“Will clinical trials go on my permanent medical record?”
Generally no. Clinical trial participation is separate from your regular medical care, though you should disclose participation to your personal doctor.
“Can I quit a study if I don’t like it?”
Absolutely. You can withdraw at any time for any reason. You’ll typically receive prorated compensation for completed portions.
“Do I need health insurance to participate?”
No. The study covers all trial-related medical care, and healthy volunteer studies don’t require insurance.
“What if I have a full-time job?”
Many outpatient studies offer evening and weekend appointments. Inpatient studies work well if you have vacation time or flexible scheduling.
The Future of Clinical Trial Participation as a Side Hustle
The clinical research industry is growing rapidly, with more trials launching each year. Fortrea’s commitment to innovation means the participant experience will only improve, with more flexible options and better compensation.
Emerging Trends
Decentralized trials: More work done from home using telehealth
Wearable technology: Continuous monitoring without facility visits
Virtual check-ins: Reducing the need for in-person appointments
Better matching algorithms: Finding the right studies faster
These innovations make clinical trials an increasingly viable option for anyone looking to build multiple income streams.
Making Your Decision About Clinical Trial Participation
Participating in Fortrea clinical trials isn’t for everyone, but it could be perfect for you if:
✅ You’re comfortable with medical procedures ✅ You can commit to study schedules ✅ You’re looking for legitimate extra income ✅ You want flexible earning opportunities ✅ You’re interested in contributing to medical research
It might not be ideal if:
❌ You have significant health issues (unless disease-specific trials) ❌ You can’t handle needles or blood draws ❌ You need guaranteed, predictable income every month ❌ You’re uncomfortable with potential side effects ❌ You can’t commit to follow protocols exactly
The key is understanding what you’re signing up for and making informed decisions about which studies align with your goals and comfort level.
Getting Started This Week
Ready to explore Fortrea clinical trials as a potential side income source? Here’s your action plan:
Today: Browse available studies in your area
This week: Submit applications for 2-3 studies you qualify for
Stay organized: Create a spreadsheet tracking studies, eligibility dates, and compensation
Plan ahead: Mark your calendar with washout periods and potential study dates
Diversify: While waiting for callbacks, explore other side hustle options to build multiple income streams
Clinical trials work best as part of a diversified side hustle portfolio, not as your sole income source. But for the right person with the right approach, Fortrea clinical trials can generate thousands of dollars per year while requiring minimal special skills or equipment.
Clinical trials offer a unique opportunity to earn substantial extra income while contributing to medical advances that could help millions. Whether you’re building your freelance career or exploring different ways to make money, Fortrea clinical trials provide legitimate, well-compensated opportunities. If you’re ready to discover more proven side hustles that fit your lifestyle and goals, Side Hustle Hackers offers honest reviews, practical resources, and community support to help you build real online income through flexible opportunities that actually work.
